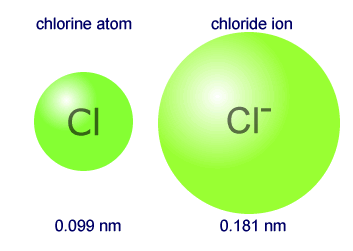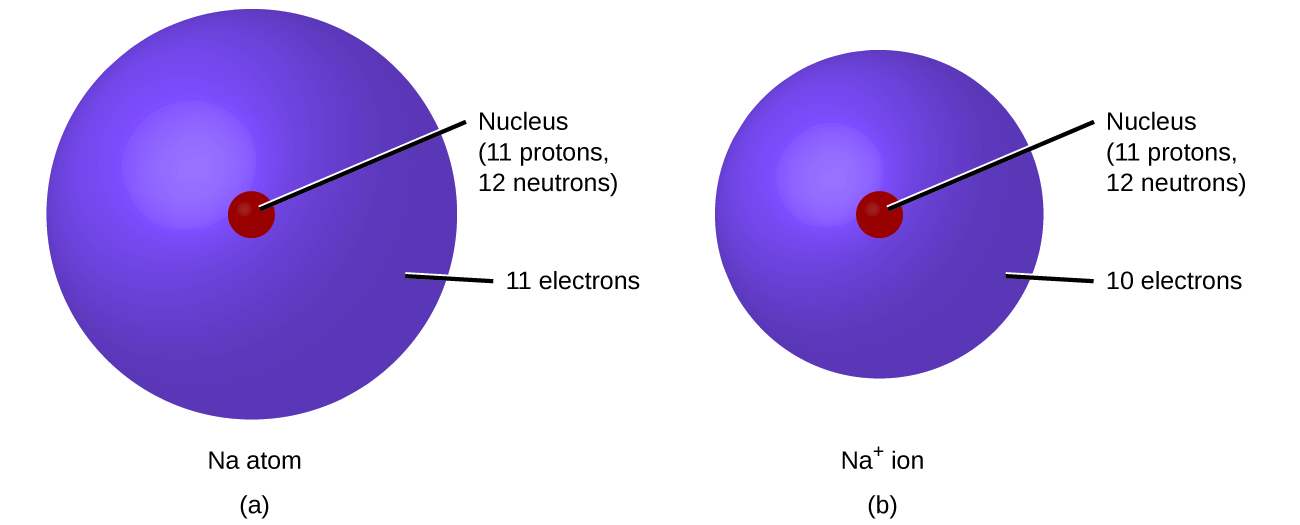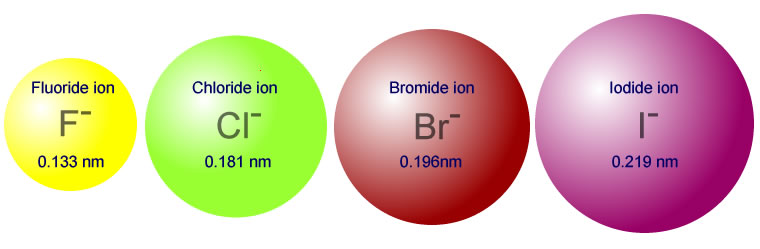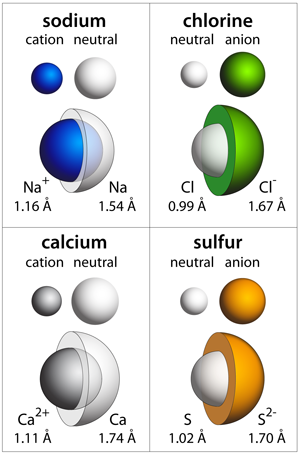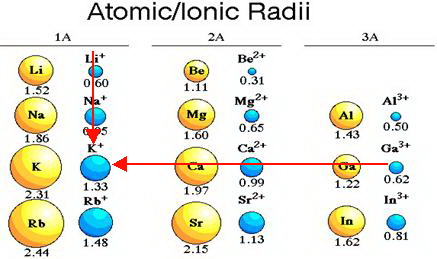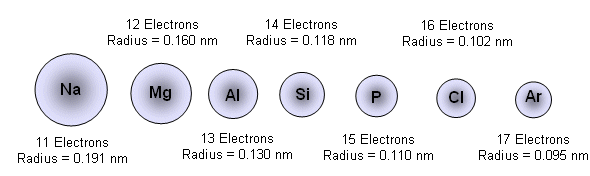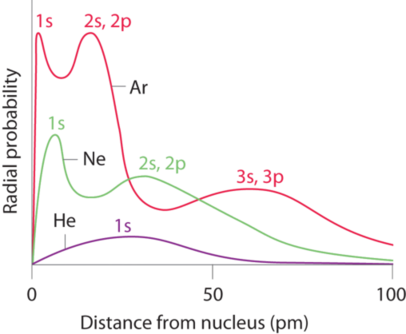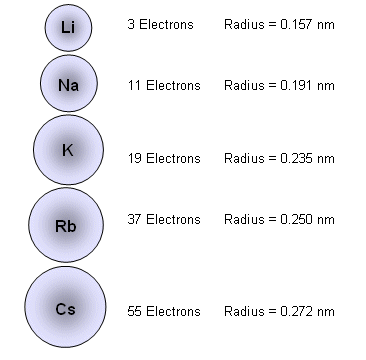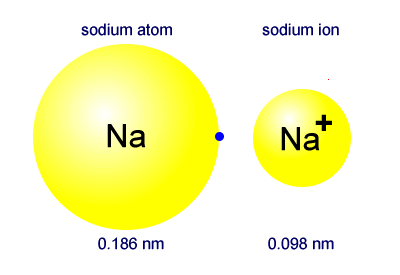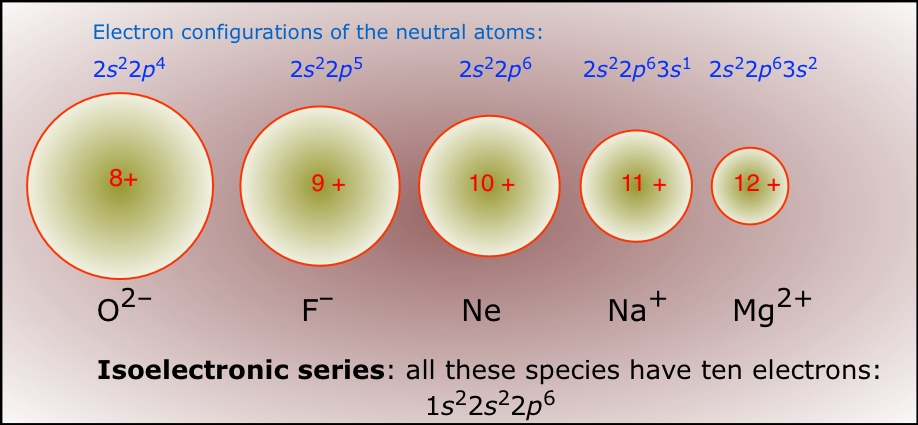
The primary reason sodium ions are smaller than sodium atoms is that the ion has only two shells of electrons (the atom has three). Some resources suggest the ion gets smaller since
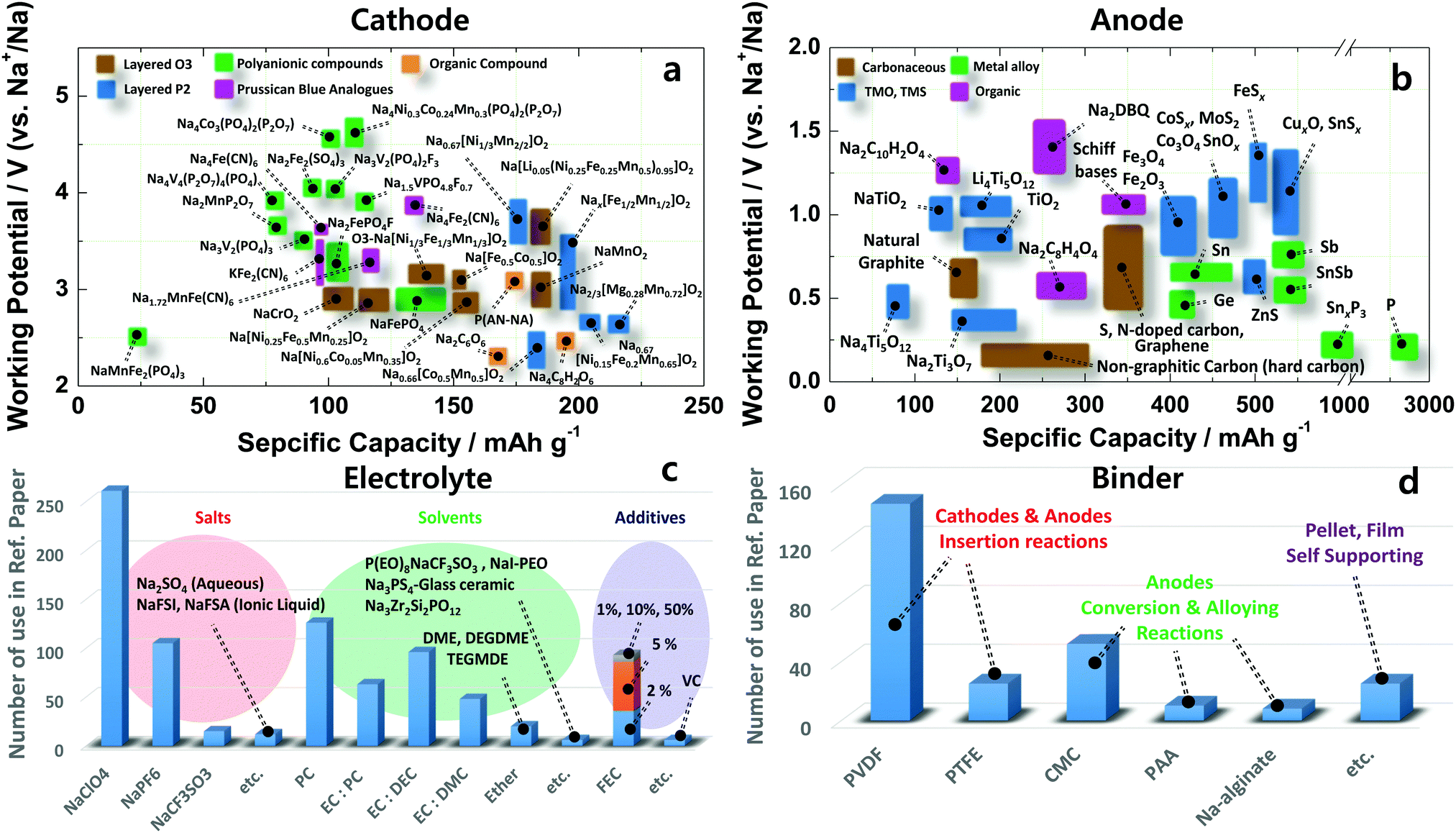
Sodium-ion batteries: present and future - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C6CS00776G

High Stability and Long Cycle Life of Rechargeable Sodium-Ion Battery Using Manganese Oxide Cathode: A Combined Density Functional Theory (DFT) and Experimental Study | ACS Applied Materials & Interfaces

Electrode Materials for Sodium-Ion Batteries: Considerations on Crystal Structures and Sodium Storage Mechanisms | SpringerLink

Structural engineering of electrode materials to boost high-performance sodium-ion batteries - ScienceDirect