One - gram mole of nitrogen occupies 2 × 10^4 cc at a pressure of 10^6 dynes/cm^2 . The average energy of a nitrogen molecule (in erg) will be: (Avogadro's number = 6 × 10^25 )
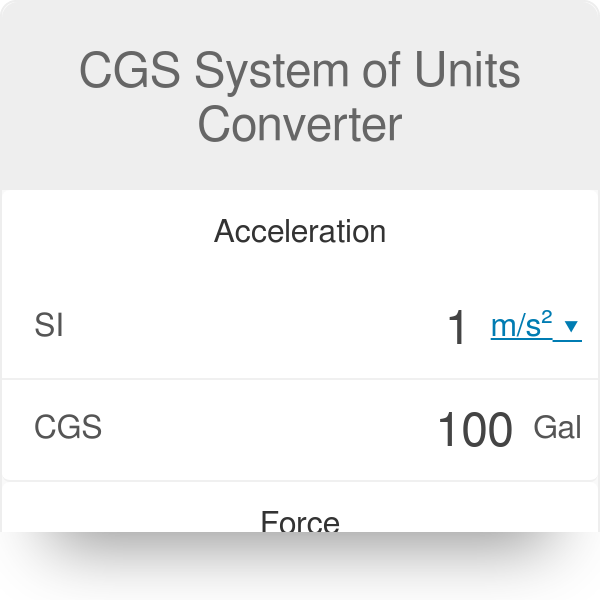
The value of Gravitional cons†an t in MKS system is 6.67×10^ 11 N m^2/kg^2. What will be its value in CGS system

SOLVED: An object of mass 100 g is at rest. A net force of 2000 dynes is applied for 10 sec. What is the final velocity? How far will the object have

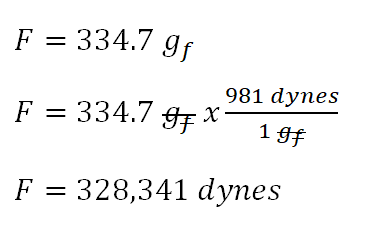

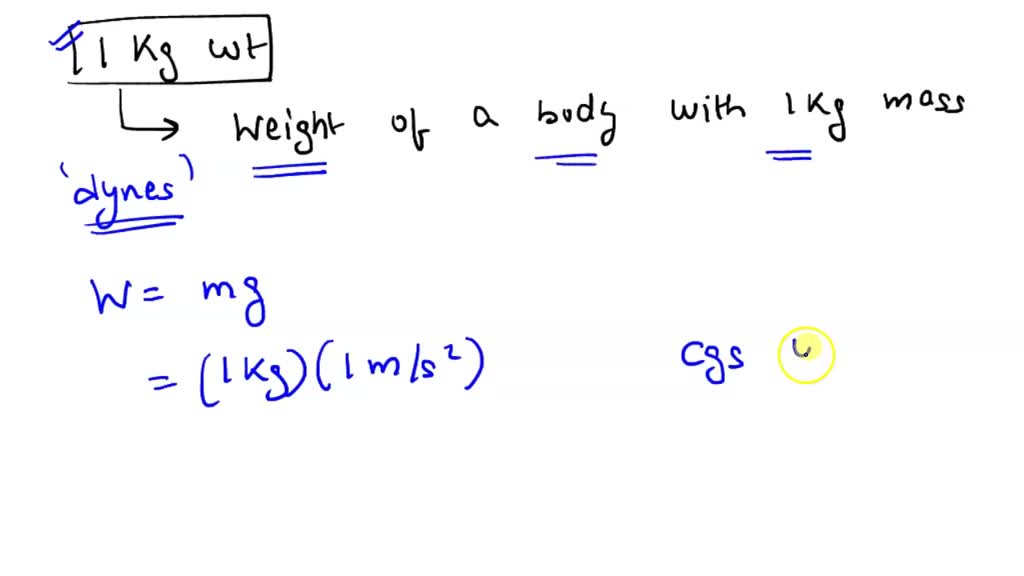

![AES E-Library » The Problems of Low-frequency [Sound] Reproduction AES E-Library » The Problems of Low-frequency [Sound] Reproduction](https://www.aes.org/images/e-lib/thumbnails/1/7/17817_full.png)